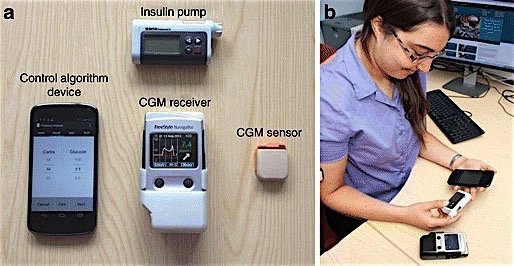
Type 1 diabetes sufferers face peculiarly onerous daily demands to maintain their health. They must frequently monitor their blood glucose levels by taking a sample of their blood, and must be constantly aware of day-to-day and within-day variability in those levels, responding by adjusting their insulin delivery. This imposes a “significant psychological burden” and can adversely affect the diabetic’s quality of life, according to researchers at Cambridge University in the UK. At present, diabetics have few alternatives, other than pancreas transplantation. But the development of an “artificial pancreas,” really a closed-loop system consisting of an algorithm-controlled insulin pump that continuously and automatically adjusts the amount of insulin entering the patient’s body, is about to change all of that.
No longer will the diabetic have to monitor his own blood sugar levels several times a day and administer insulin separately. By joining the two actions together in a closed loop, Dr. Roman Hovorka and Dr. Hood Thabit of Cambridge University say they can give diabetics “time off” from their diabetes management, relieving them of the constant concern about becoming either hyperglycaemic (too much blood sugar) or the opposite, hypoglycaemic. Their artificial pancreas reduces the burden of self-care while improving glucose control.
The average diabetic’s insulin requirement varies by 20 per cent during waking hours and 30 per cent at night, but the variability can be even greater depending on diet, exercise and other factors. Unlike conventional insulin pumps, a closed-loop system like this one combines real-time sensors to measure glucose levels with autonomous delivery of insulin as needed. In the Cambridge researchers’ prototype system, the control algorithm runs on a mobile phone. The other components are the continuous glucose monitor and receiver, and the insulin pump.
Responding to day-to-day and within-day glucose variability is the key advantage that adaptive closed-loop systems have over conventional insulin therapies. By autonomously and continually modulating insulin delivery in a glucose-responsive fashion, closed-loop systems deliver insulin to minimise hyper- and hypoglycaemic excursions. Adaptive features of the control algorithms individualise the closed-loop system to particular physiology and lifestyle patterns.
The closed loop device also eliminates the need for the major surgery of a pancreatic transplant, and the subsequent need for immunosuppressant drugs.
The authors report that clinical trials have been carried out in various settings, including controlled laboratory conditions, diabetes camps, and outpatient settings. Trials have shown “as good or better” glucose control than existing technologies. Trials are ongoing, and more studies are planned for both adults and children. Home studies are the “final benchmark” testing environment, and unsupervised free-living home studies have been performed with good results, the researchers say.
At least $200 million has gone into the effort to develop the artificial pancreas over the past decade. The device could receive approval from the US Food and Drug Administration as early as 2017 and could be on the market by 2018.